Eylea is a medication used for the treatment of certain eye conditions, particularly retinal diseases characterized by abnormal blood vessel growth. The active ingredient in Eylea is aflibercept.
Aflibercept is a vascular endothelial growth factor (VEGF) inhibitor. VEGF is a protein that plays a crucial role in promoting the growth of new blood vessels, a process known as angiogenesis. In certain eye conditions, such as wet age-related macular degeneration (AMD), diabetic macular edema (DME), and macular edema following retinal vein occlusion (RVO), abnormal blood vessel growth can lead to vision problems.
Eylea works by blocking the effects of VEGF, thereby inhibiting the growth of abnormal blood vessels and reducing vascular permeability. This helps to alleviate the underlying causes of vision impairment in these conditions.
Conditions for which Eylea is commonly prescribed include:
- Wet Age-Related Macular Degeneration (AMD): Eylea is approved for the treatment of wet AMD, a condition where abnormal blood vessels grow beneath the retina, leading to vision loss.
- Diabetic Macular Edema (DME): Eylea is used to treat DME, a complication of diabetic retinopathy characterized by fluid accumulation in the macula, leading to vision problems.
- Macular Edema Following Retinal Vein Occlusion (RVO): Eylea is indicated for the treatment of macular edema resulting from central retinal vein occlusion (CRVO) or branch retinal vein occlusion (BRVO).
Eylea is typically administered through injections directly into the eye (intravitreal injections). The frequency and duration of treatment are determined by the prescribing healthcare professional based on the specific eye condition and individual patient response.
As with any medication, Eylea may have potential side effects, and its use should be carefully monitored by an ophthalmologist or retinal specialist. Patients considering or undergoing treatment with Eylea should discuss any concerns or questions with their healthcare provider.
High dose Eylea was approved in 2023 for the treatment of wet macular degeneration and diabetic macular edema. HD Eylea is a four times higher dose of drug per injection compared to regular Eylea.
EYLEA HD helped 83% of patients with Wet AMD and 93% with DME go 3 or 4 months between treatments after 3 initial monthly doses.
Eylea (aflibercept) is a medication that has been found to be effective in the treatment of wet age-related macular degeneration (AMD), providing several benefits for patients with this condition. Here are some details on the benefits of using Eylea for wet AMD:
- Inhibition of Abnormal Blood Vessel Growth: Wet AMD is characterized by the growth of abnormal blood vessels beneath the retina, a process known as choroidal neovascularization. Aflibercept, the active ingredient in Eylea, is a vascular endothelial growth factor (VEGF) inhibitor. It works by blocking the effects of VEGF, a protein that promotes the growth of new blood vessels. By inhibiting VEGF, Eylea helps to prevent the formation of these abnormal blood vessels, reducing the leakage and bleeding that can occur in the retina.
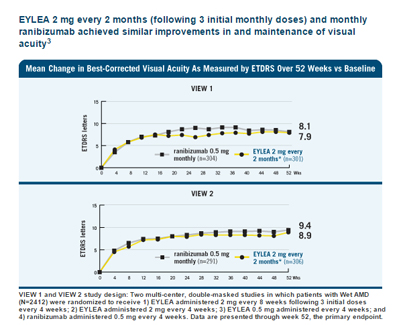
- Improvement in Visual Acuity: Clinical trials and real-world studies have demonstrated that Eylea can lead to significant improvements in visual acuity for many patients with wet AMD. By targeting the underlying cause of vision loss—the abnormal blood vessel growth—Eylea can help stabilize or even improve vision in some cases.
- Reduced Central Retinal Thickness: Eylea has been shown to reduce central retinal thickness, which is often increased in wet AMD due to fluid accumulation and swelling. By addressing macular edema (swelling in the central part of the retina), Eylea contributes to improved retinal structure and function.
- Extended Treatment Intervals: Eylea has demonstrated a longer duration of action compared to some other anti-VEGF medications. This means that, in some cases, patients may require less frequent injections, potentially reducing the burden of treatment on both patients and healthcare providers.
- Maintenance of Visual Gains Over Time: Long-term studies have indicated that visual gains achieved with Eylea treatment can be maintained over an extended period. This suggests the sustained effectiveness of the medication in managing wet AMD.
It’s important to note that while Eylea has shown significant benefits for many individuals with wet AMD, individual responses to treatment can vary. The decision to use Eylea, as well as the specific treatment plan, should be made in consultation with an ophthalmologist or retinal specialist who will consider the patient’s overall health and the characteristics of their eye condition. Regular follow-up appointments and monitoring are typically part of the treatment plan to assess the ongoing effectiveness of Eylea.
Eylea (aflibercept) is also used in the treatment of macular edema following central retinal vein occlusion (CRVO) and branch retinal vein occlusion (BRVO). Here are some additional details on the use of Eylea for central retinal vein occlusion:
- Mechanism of Action: Similar to its use in wet age-related macular degeneration (AMD), Eylea acts as a vascular endothelial growth factor (VEGF) inhibitor. In the context of retinal vein occlusion, abnormal blood vessel growth and increased vascular permeability can lead to macular edema, causing vision impairment. By blocking VEGF, Eylea helps to reduce the growth of abnormal blood vessels and decrease fluid leakage, thus addressing macular edema associated with CRVO and BRVO.
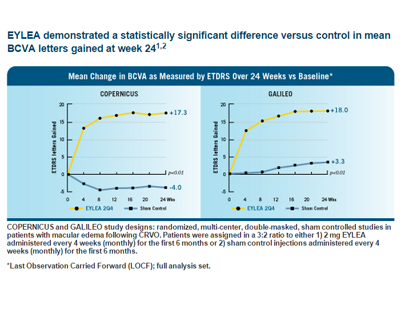
- Macular Edema Resolution: Eylea has been shown to effectively reduce macular edema, which is the accumulation of fluid in the macula, the central part of the retina. This reduction in edema contributes to an improvement in visual acuity for some patients with CRVO and BRVO.
- Visual Acuity Improvement: Clinical studies and real-world evidence have demonstrated that Eylea treatment can lead to improvements in visual acuity in individuals with macular edema following CRVO and BRVO. The degree of improvement can vary among patients.
- Treatment Schedule: The treatment schedule for Eylea in CRVO and BRVO typically involves an initial loading phase of monthly injections, followed by a maintenance phase with less frequent injections. The specific treatment plan is determined by the treating ophthalmologist based on the patient’s response to the medication.
- Reduced Central Retinal Thickness: Eylea has been shown to reduce central retinal thickness, indicating a reduction in macular edema. This reduction is associated with improvements in retinal structure and function.
- Extended Treatment Intervals: Similar to its use in wet AMD, Eylea has demonstrated a longer duration of action in some cases, allowing for extended treatment intervals. This may contribute to reduced treatment burden for patients.
In general, about 40 percent of patients with branch retinal vein occlusion and 60 percent of patients with central retinal vein occlusion require more than four years of therapy with Eylea.
Eylea (aflibercept) is approved for the treatment of diabetic macular edema (DME), a condition that can occur as a complication of diabetic retinopathy. Here are more details on the use of Eylea for diabetic macular edema:
- Mechanism of Action: Eylea is a vascular endothelial growth factor (VEGF) inhibitor. In DME, increased levels of VEGF contribute to abnormal blood vessel growth and increased vascular permeability, leading to fluid leakage and swelling in the macula (central part of the retina). By blocking VEGF, Eylea helps to reduce the growth of abnormal blood vessels and decrease fluid leakage, addressing the underlying causes of DME.
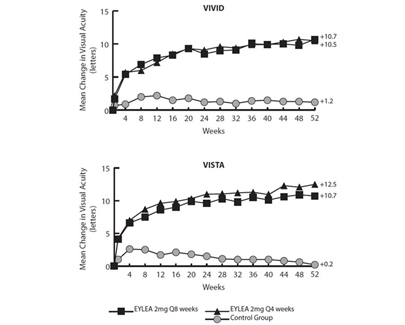
- Improvement in Visual Acuity: Clinical studies have shown that Eylea treatment can lead to significant improvements in visual acuity for many patients with DME. By reducing macular edema and addressing the associated vision problems, Eylea helps improve central vision.
- Reduction in Central Retinal Thickness: Eylea has been demonstrated to reduce central retinal thickness, which is often increased in DME due to fluid accumulation. The reduction in macular thickness contributes to improvements in retinal structure and function.
- Treatment Schedule: The treatment schedule for Eylea in DME typically involves an initial loading phase of monthly injections, followed by a maintenance phase with less frequent injections. The specific treatment plan is determined by the treating ophthalmologist based on the patient’s response to the medication.
- Extended Treatment Intervals: Similar to its use in other retinal conditions, Eylea has shown a longer duration of action in some cases, allowing for extended treatment intervals. This can be beneficial in reducing the frequency of injections and the overall treatment burden for patients.
- Combined Therapy: In some cases, Eylea may be used in combination with other treatment modalities, such as laser therapy, to optimize the management of DME. The combination approach is tailored to the individual patient’s needs and the characteristics of their DME.
- Safety Profile: Eylea has demonstrated a generally favorable safety profile in clinical trials for DME. As with any medical treatment, potential risks and benefits should be discussed with the treating healthcare provider.
Everyone is different, but in general most patients with diabetic macular edema require about 10-12 injections the first year, about 6-8 injections the second year and then about 2-4 injections the third year of treatment. Many but not all patients with diabetic macular edema do not require ongoing treatment with intravitreal injections.